Patient safety remains the foremost priority in pharmaceutical manufacturing, driving the implementation of sophisticated inspection technologies that work in concert to ensure final product integrity. This integrated approach combines metal detection, weight verification, visual inspection, and X-ray examination to create a comprehensive quality assurance ecosystem. Each technology serves a distinct yet complementary role in safeguarding product quality throughout the manufacturing journey.
The quality assurance process begins at the material preparation stage, where raw powders are milled, sifted, and blended before granulation. At this critical point, a free-fall metal detector inspects the granules as they descend through a vertical chute.
The system is equipped with a specialized small-aperture coil configuration that provides exceptional sensitivity for detecting stainless steel contaminants—often originating from equipment wear in previous processing steps.

Furthermore, its robust design withstands environmental challenges like vibration and electromagnetic interference, while integrated failsafe mechanisms guarantee the automatic rejection of any contaminated material during system anomalies.
This initial inspection phase is fundamental to the quality strategy, preventing metallic contaminants from propagating further into the production process.
The examination process continues with weight verification of empty capsule shells in their pre-locked state. A specialized checkweigher utilizing electromagnetic force restoration technology delivers exceptional accuracy within ±0.3 g, ensuring shell consistency essential for subsequent dosage precision. The system's stable feeding mechanism prevents product bouncing and maintains measurement integrity throughout operation.

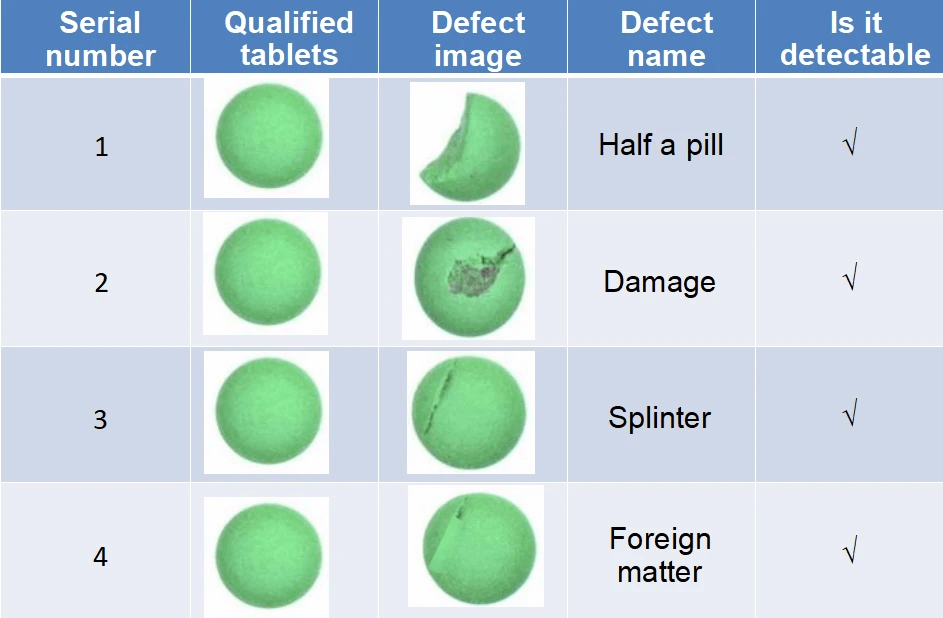
Following formation and filling operations, products undergo rigorous visual examination through tablet automated inspection systems. These advanced platforms employ multiple high-resolution cameras to capture detailed imagery from all perspectives, including top, bottom, and side surfaces. Sophisticated algorithms analyze these images to identify various imperfections such as chips, cracks, surface stains, scratches, discoloration, and marking irregularities.
Operating at speeds up to 200,000 capsules/min, these systems provide consistent and reliable inspection capabilities that far surpass manual methods. This automated approach ensures uniform quality assessment while eliminating human subjectivity and fatigue factors.
Products subsequently pass through a gravity-fed metal detection system equipped with anti-static chuting to eliminate false signals caused by static electricity. This additional inspection stage offers supplementary protection against metallic contamination.
The weight assessment process completes with high-speed multi-lane checkweighers processing approximately 230,000 units hourly. This ultimate verification step confirms dosage accuracy, representing an essential factor for therapeutic effectiveness and patient safety.
The validation process reaches its conclusion with X-ray inspection of final packaged products. These systems provide superior detection capabilities, identifying diverse foreign materials including various metals, glass fragments, bone particles, and certain plastic contaminants within finished products. Furthermore, they conduct essential package integrity assessment, detecting missing, damaged, or deformed units through packaging materials.
At LTPM, we engineer and manufacture complete pharmaceutical production systems—from high-precision capsule filling machines and tablet presses to comprehensive inspection equipment—all designed to work together seamlessly. Our integrated approach ensures unmatched quality control throughout your manufacturing process.
Transform your production line with LTPM's end-to-end solutions. Contact us today to discover how our customized equipment portfolio can optimize your operations, ensure regulatory compliance, and deliver uncompromising product quality.
Contact us via [email protected] to schedule a consultation with our pharmaceutical equipment specialists.
We use cookies to enhance your browsing experience and protect your privacy. Please read our privacy policy for more information.